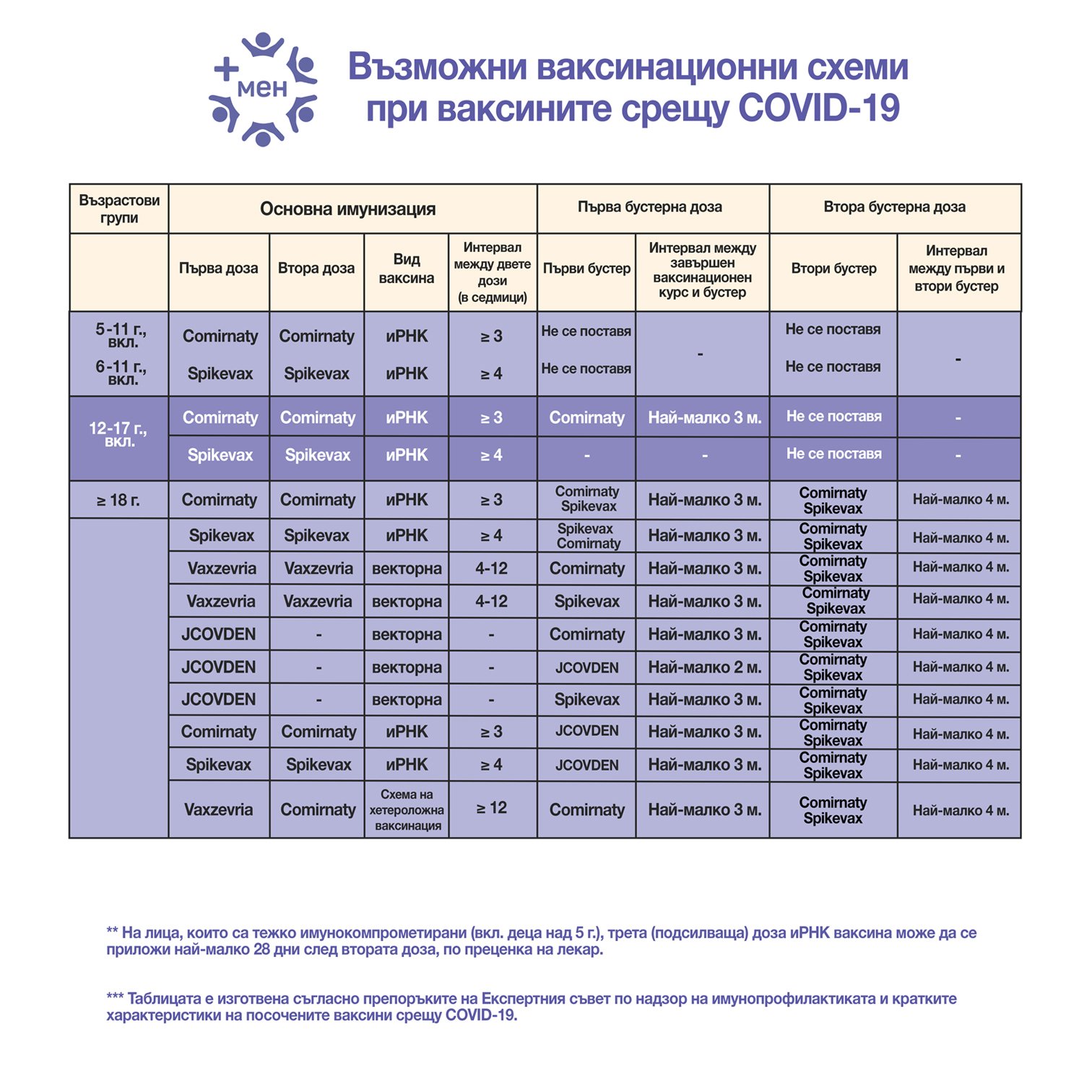
Ministry of Health has approved the recommendations of the Expert Council on Immunoprophylaxis Supervision regarding the administration of a fourth dose (second booster) of mRNA vaccine against COVID-19 for all individuals aged 18 and over who wish to receive it. The second booster dose can be administered at least 4 months after the first booster to individuals whose completed vaccination course is with a two-dose vaccine.
The second booster dose of mRNA vaccine is highly recommended for the following categories of individuals aged 18 and over:
- Immunocompromised individuals;
- Transplant patients receiving immunosuppressive therapy;
- Patients on chronic dialysis;
- Patients with oncohematological diseases;
- Residents and staff in social institutions providing long-term care for the elderly;
- Medical professionals involved in the treatment of COVID-19 patients;
- Elderly individuals aged 65 and over.
The Expert Council's opinion is in line with the recommendation of the European Medicines Agency and the European Centre for Disease Prevention and Control to ensure that at-risk individuals in the population can receive a fourth dose (second booster) of mRNA vaccine against COVID-19.
The second booster vaccine against COVID-19 can be administered at the vaccination points of the Regional Health Inspectorates, the offices of general practitioners, as well as in medical institutions for hospital and outpatient medical care.
As of Monday, June 27, the National Immunization Register – part of the National Health Information System – will allow the record of a second booster mRNA vaccine administered, and citizens will also receive a vaccination certificate. Information Service is already working on adapting the system's functionalities.
The certificate will be identical to that for the first booster dose, with the only exception being the numbering of the administered vaccine – for the second booster vaccine, the certificate will indicate 4/4. This certificate, like the certificate for the first booster vaccine, will be indefinite, as long as the current European regulation does not provide otherwise.